Role of Cytogenetics and Molecular Cytogenetics in Diagnosing Genetic Imbalances
Introduction
Over the past five decades, cytogenetics and molecular cytogenetics have transformed the diagnosis of genetic imbalances associated with developmental delay and congenital abnormalities. These technologies enable the detection of chromosomal alterations ranging from large-scale numerical abnormalities to subtle microdeletions and duplications. Today, integrating conventional cytogenetic methods with advanced molecular tools significantly improves diagnostic accuracy and patient management.
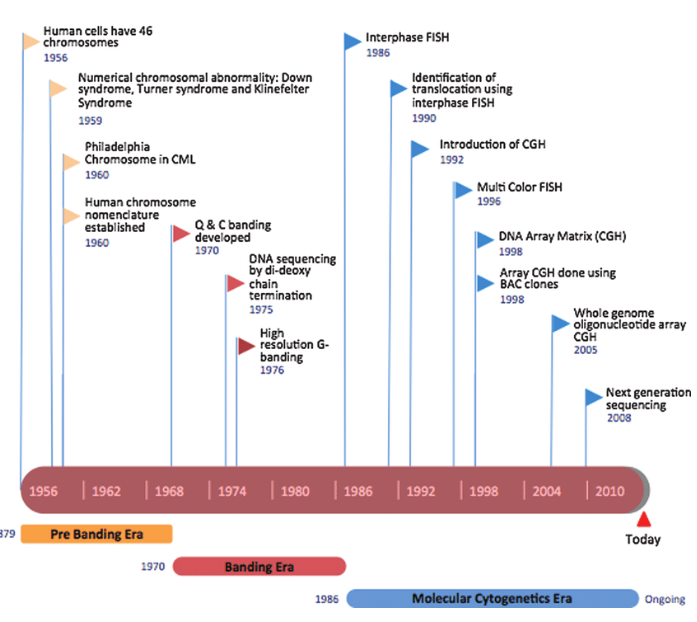 Evolution of Cytogenetic Techniques
Evolution of Cytogenetic Techniques
Initially, cytogenetics focused on identifying numerical chromosomal abnormalities such as aneuploidy. The introduction of chromosome banding techniques in the 1970s marked a major breakthrough, allowing precise identification of individual chromosomes and structural abnormalities, including deletions, duplications, and rearrangements linked to specific syndromes.
Subsequent innovations, particularly fluorescence in situ hybridization (FISH) and comparative genomic hybridization (CGH) technologies, expanded diagnostic capabilities by enabling detection of submicroscopic genomic alterations that were previously undetectable.
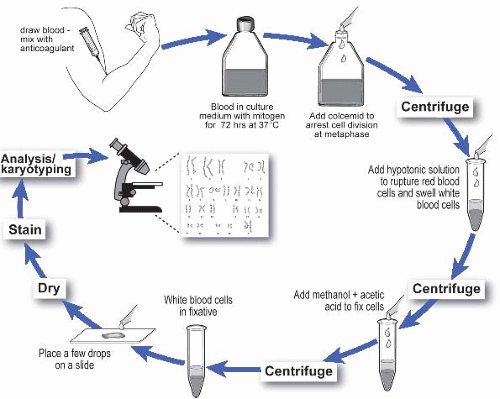
Classic Cytogenetic Analysis
Principle and Clinical Relevance

Conventional cytogenetic analysis, also known as karyotyping, remains the first-line diagnostic test for patients presenting with:
- Developmental delay
- Multiple congenital anomalies
- Suspected chromosomal disorders
A high-resolution karyotype (≥650 bands) is recommended to detect subtle chromosomal changes. Typically, at least 20 metaphase cells are analyzed to ensure accurate detection of:
- Aneuploidy
- Structural abnormalities
- Chromosomal mosaicism
Advantages
- Detects large-scale chromosomal abnormalities
- Identifies balanced rearrangements
- Useful for initial screening
Limitations
- Limited resolution for small deletions or duplications
- May miss submicroscopic genomic imbalances
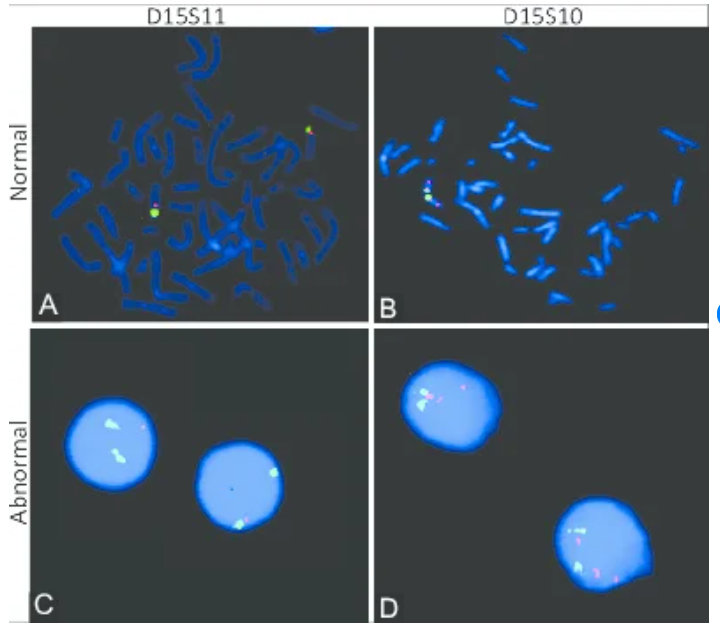
Fluorescence In Situ Hybridization (FISH)
FISH is a targeted molecular cytogenetic technique that uses labeled DNA probes to detect specific chromosomal regions. It is highly effective for identifying known or suspected genetic abnormalities.
Types of FISH Probes
- Alpha-satellite probes: Detect centromeric regions (useful for aneuploidy)
- Beta-satellite probes: Analyze acrocentric chromosome regions
- Telomeric/subtelomeric probes: Identify terminal deletions or duplications
- Gene-specific probes: Detect microdeletion and microduplication syndromes
- Whole chromosome painting probes: Determine chromosomal origin of rearrangements
Applications
- Rapid detection of aneuploidy
- Identification of specific syndromes (DiGeorge syndrome, Williams syndrome)
- Characterization of marker or derivative chromosomes
Limitations
FISH is highly specific but not genome-wide, meaning it requires prior clinical suspicion and cannot efficiently screen the entire genome.
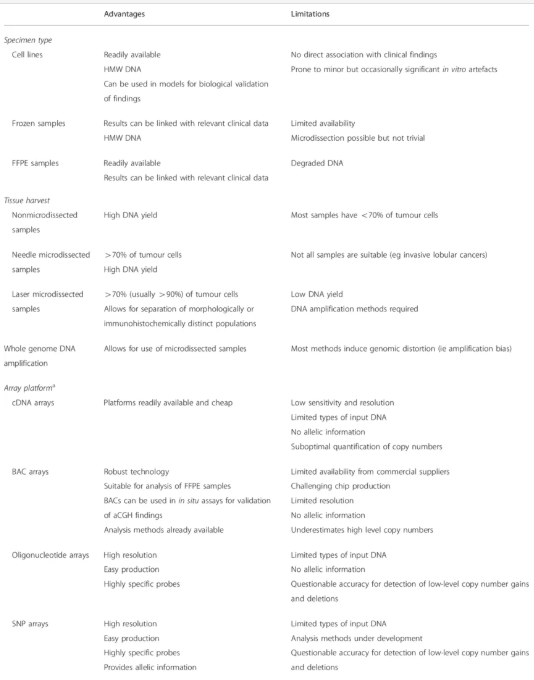
Array Comparative Genomic Hybridization (aCGH)
Technology Overview
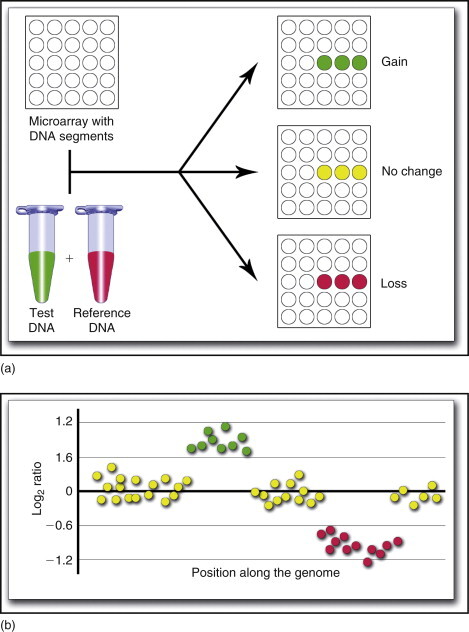
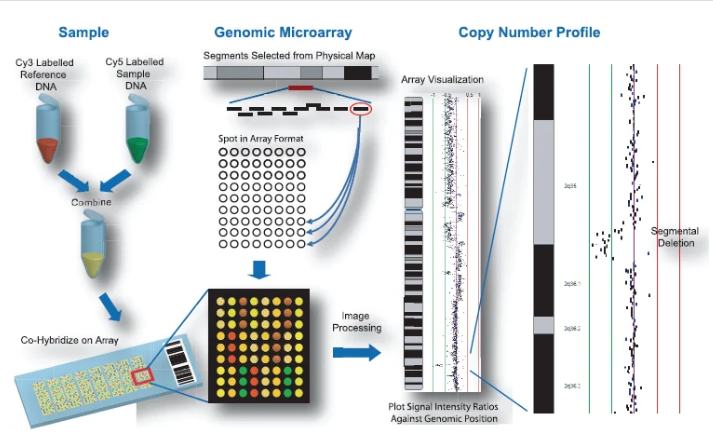
Array CGH (aCGH) represents a high-resolution, genome-wide screening method for detecting copy number variations (CNVs), including microdeletions and microduplications.
This technique compares patient DNA with control DNA to identify genomic gains or losses across hundreds to thousands of loci simultaneously.
Advantages
- Genome-wide analysis in a single assay
- High sensitivity for submicroscopic abnormalities
- Detection of previously unknown chromosomal imbalances
- Superior resolution compared to karyotyping and FISH
Limitations
- Cannot reliably detect balanced rearrangements
- May identify benign copy number variants (polymorphisms)
- Requires confirmation (often by FISH)
- Interpretation depends on genomic databases
Clinical Impact
In patients with developmental delay and normal karyotypes, aCGH increases the detection rate of genomic imbalances by approximately 8%, identifying clinically relevant abnormalities that conventional methods miss.
Common Microdeletion and Microduplication Syndromes
Several clinically significant syndromes are associated with specific chromosomal regions, including:
- 1p36 deletion syndrome
- Wolf-Hirschhorn syndrome (4p16.3)
- Cri-du-Chat syndrome (5p15.2)
- Williams syndrome (7q11.23)
- Prader-Willi / Angelman syndromes (15q11.2)
- DiGeorge syndrome (22q11.2)
- Duchenne muscular dystrophy (Xp21.2)
These conditions highlight the importance of high-resolution genomic analysis for accurate diagnosis.
Diagnostic Strategy in Clinical Practice
A structured diagnostic approach improves efficiency and accuracy:
Step 1: Initial Testing
- Perform high-resolution cytogenetic analysis
Step 2: Targeted Testing
- Use FISH when a specific syndrome is suspected
Step 3: Genome-Wide Screening
- Apply aCGH if:
- Cytogenetic results are anormal
- Phenotype suggests an unknown chromosomal imbalance
Step 4: Confirmation and Refinement
- Confirm findings with FISH
- Use combined approaches to refine breakpoints and gene involvement
Clinical Considerations
- Some disorders are caused by gene mutations rather than copy number changes, requiring molecular genetic testing instead of cytogenetic analysis
- Certain conditions ( Fragile X syndrome, metabolic disorders) are not detectable by FISH or aCGH
- Tissue selection is critical, as some mosaic conditions may not be detectable in blood samples
Conclusion
The integration of classic cytogenetics, FISH, and aCGH has revolutionized the detection of genomic imbalances. While conventional cytogenetics remains essential for initial screening, molecular cytogenetic techniques significantly enhance diagnostic resolution and uncover hidden abnormalities.
A combined, stepwise diagnostic strategy ensures optimal identification of chromosomal disorders, ultimately improving clinical decision-making and patient care.
Recent Posts
-
Randomly Amplified Polymorphic DNA (RAPD)
Introduction The analysis of genetic variation at the DNA level has become a fundamental approach in …1st Apr 2026 -
Role of Cytogenetics and Molecular Cytogenetics in Diagnosing Genetic Imbalances
Introduction Over the past five decades, cytogenetics and molecular cytogenetics have transformed t …1st Apr 2026 -
In Situ Hybridization in Cytogenetics
Introduction In situ hybridization (ISH) is a powerful technique used in cytogenetics to detect and …31st Mar 2026