Genome of Anopheles gambiae: What We Know Today
Genome of Anopheles gambiae: What We Know Today
Mosquitoes are among the most dangerous animals on Earth not because of their bite itself, but because of the pathogens they transmit. Among them, Anopheles gambiae stands out as one of the most efficient vectors of malaria, responsible for a significant proportion of cases in sub-Saharan Africa. Its success as a vector is not accidental it is deeply rooted in its genetic makeup. Over the past two decades, genomic research has transformed our understanding of this species, revealing how its DNA encodes behaviors, physiological traits, and adaptations that make it highly specialized for human infection cycles. Studying its genome is therefore essential not only for basic science, but also for designing new strategies to combat malaria.
A Milestone: Sequencing the Mosquito Genome
The sequencing of the Anopheles gambiae genome in 2002 marked a turning point in vector biology and infectious disease research. For the first time, scientists had access to the complete genetic blueprint of a major malaria vector, opening the door to large-scale functional studies. The genome, consisting of approximately 280 million base pairs and over 13,000 genes, may appear modest compared to the human genome, but it is highly specialized. Many of its genes are dedicated to survival in specific ecological niches, including blood feeding, reproduction, and environmental adaptation. This sequencing effort also enabled comparative genomics, allowing researchers to identify differences between mosquito species and understand why Anopheles gambiae is particularly efficient at transmitting malaria. Since then, continuous improvements in sequencing technologies have refined the genome, uncovering regulatory elements and structural variations that further explain its biological success.
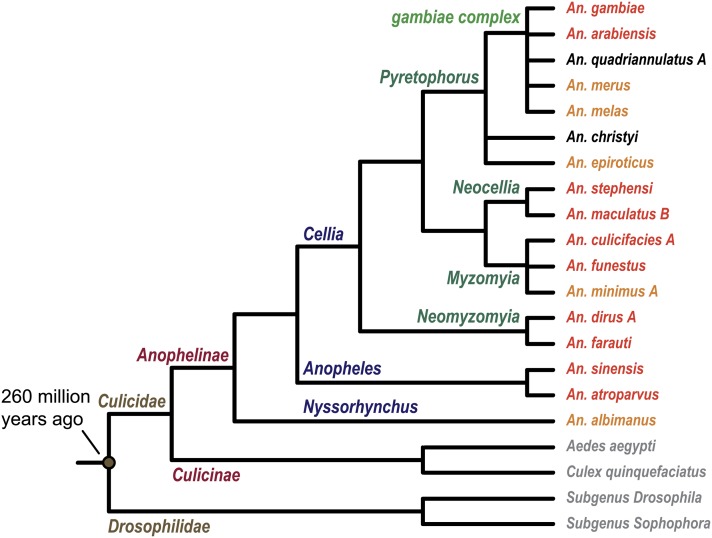
Figure : Illustration of the 16 anophelines and their relationships to An. gambiae, the two sequenced culicines Aedes aegypti and Culex quinquefasciatus and the sequenced Drosophila species.
Genes That Drive Malaria Transmission
The ability of Anopheles gambiae to transmit malaria depends on a complex network of genes that regulate its interaction with both humans and the malaria parasite. Unlike other mosquitoes that may feed on a variety of animals, this species has evolved genetic traits that strongly favor human hosts. These traits are not controlled by a single gene but by coordinated expression of multiple gene families that influence behavior, physiology, and parasite compatibility. Understanding these genes is crucial because even small genetic variations can significantly alter transmission efficiency. By identifying and studying these genetic pathways, researchers can pinpoint vulnerabilities in the mosquito’s biology that could be exploited to interrupt the malaria transmission cycle.
1. Host-Seeking and Blood Feeding
One of the defining features of Anopheles gambiae is its remarkable ability to locate human hosts. This behavior is driven by a sophisticated set of olfactory and sensory genes that detect carbon dioxide, lactic acid, and other volatile compounds emitted by human skin. These olfactory receptor genes are highly diversified and finely tuned, allowing the mosquito to distinguish humans from other animals even in complex environments. In addition, genes involved in visual and thermal sensing help the mosquito track movement and body heat, increasing its chances of successful feeding. Blood feeding itself is also genetically regulated, involving salivary proteins that prevent blood clotting and reduce host immune responses. Together, these genetic adaptations make Anopheles gambiae exceptionally efficient at finding and feeding on humans, directly contributing to its role as a malaria vector.
2. Immune System and Parasite Interaction
Once a mosquito ingests blood containing Plasmodium parasites, a critical battle begins inside its body. The mosquito’s immune system determines whether the parasite will be destroyed or allowed to develop further. This immune response is controlled by several gene families, including pattern recognition receptors, signaling molecules, and effector proteins such as antimicrobial peptides. Some mosquitoes possess genetic variants that enhance their ability to kill the parasite, while others are more permissive, allowing the parasite to complete its life cycle. This variation in “vector competence” is a key area of research, as it influences transmission dynamics at the population level. By understanding how these immune genes function and interact, scientists hope to develop strategies that enhance the mosquito’s natural resistance to Plasmodium, thereby reducing disease transmission.
3. Salivary Gland Proteins
For malaria transmission to occur, the parasite must successfully migrate to and invade the mosquito’s salivary glands, a process that involves precise molecular interactions. Specific mosquito proteins, such as Saglin, interact with parasite proteins like TRAP, facilitating the attachment and entry of the parasite into the gland. This step represents a critical bottleneck in the transmission cycle, as failure at this stage prevents the parasite from being transmitted to a new human host. Genomic studies have identified several salivary gland proteins involved in this process, many of which are now being investigated as potential targets for intervention. By disrupting these interactions, it may be possible to block transmission without affecting the mosquito’s survival, offering a more ecologically balanced approach to disease control.
Insecticide Resistance: A Growing Genetic Challenge
Insecticide resistance has emerged as one of the greatest threats to malaria control efforts, and genomics has been instrumental in uncovering its underlying mechanisms. Resistance often arises through mutations in genes that alter the target site of insecticides, such as the well-known kdr mutations in voltage-gated sodium channels, which reduce the effectiveness of pyrethroids. In addition to these target-site changes, mosquitoes can develop metabolic resistance by overexpressing detoxification enzymes, including cytochrome P450s, glutathione S-transferases, and esterases. These enzymes break down insecticides before they can exert their toxic effects. The rapid spread of these resistance genes across mosquito populations highlights the evolutionary adaptability of Anopheles gambiae. Understanding these genetic mechanisms is essential for developing new control strategies that can overcome or bypass resistance.
Population Genomics: Tracking Mosquito Evolution
Advances in high-throughput sequencing have enabled researchers to move beyond individual genomes and study entire mosquito populations. Population genomics provides insights into genetic diversity, gene flow, and evolutionary dynamics across different regions. For Anopheles gambiae, these studies have revealed a highly structured population with significant local adaptations, driven by environmental factors and human interventions such as insecticide use. By tracking the spread of specific genes particularly those associated with resistance scientists can monitor how mosquito populations respond to control measures in real time. This information is invaluable for designing targeted interventions and predicting future trends in malaria transmission. Population genomics thus represents a powerful tool for bridging the gap between laboratory research and real-world applications.
Genomics and the Future of Mosquito Control
The growing understanding of the Anopheles gambiae genome is paving the way for innovative approaches to mosquito control. Rather than relying solely on traditional methods, researchers are now exploring strategies that target the mosquito at the genetic level. These approaches aim to reduce transmission efficiency, limit population growth, or alter mosquito behavior in ways that decrease human contact. Genomics provides the foundation for these innovations by identifying key genes and pathways that can be manipulated. As a result, mosquito control is shifting from a purely chemical approach to a more precise, biology-driven strategy.
1. CRISPR and Gene Editing
Gene editing technologies, particularly CRISPR-Cas9, have revolutionized the field by enabling precise modifications of the mosquito genome. Scientists can now introduce changes that reduce fertility, impair parasite development, or spread desirable traits through populations using gene drives. These approaches hold immense potential for long-term malaria control, as they could reduce or even eliminate the ability of mosquitoes to transmit the disease. However, they also raise important ethical and ecological questions, particularly regarding the irreversible nature of some genetic modifications. Careful evaluation and regulation will be essential before these technologies can be deployed in the field.
2. New Insecticide Targets
Genomic research is also helping identify new molecular targets for insecticide development. By understanding which genes are essential for mosquito survival or reproduction, scientists can design compounds that specifically disrupt these functions. This targeted approach could lead to more effective and environmentally friendly insecticides, reducing the impact on non-target species. Additionally, identifying genes involved in resistance allows researchers to develop strategies that either avoid or counteract these mechanisms, extending the lifespan of existing control tools.
3. Transmission-Blocking Strategies
Another promising application of genomics is the development of transmission-blocking strategies that interfere with the parasite rather than the mosquito. By targeting genes involved in parasite development or migration, it may be possible to prevent malaria transmission without reducing mosquito populations. This approach could minimize ecological disruption while still achieving significant public health benefits. Research in this area is ongoing, with several candidate genes and pathways already identified as potential targets.
Challenges and Knowledge Gaps
Despite significant progress, many aspects of the Anopheles gambiae genome remain poorly understood. A large number of genes still lack clear functional annotation, and the interactions between different genetic pathways are often complex and context-dependent. Additionally, environmental factors can influence gene expression, adding another layer of complexity to the system. Predicting how mosquito populations will evolve in response to new interventions remains a major challenge. Addressing these knowledge gaps will require continued research, interdisciplinary collaboration, and the integration of genomic data with ecological and epidemiological studies.
https://pmc.ncbi.nlm.nih.gov/articles/PMC1448890/
Conclusion
The genome of Anopheles gambiae has fundamentally changed our understanding of malaria transmission and vector biology. It reveals how a relatively small set of genes can produce a highly specialized and adaptable organism capable of sustaining one of the world’s most devastating diseases. At the same time, this knowledge provides powerful tools for developing new control strategies, from gene editing to targeted insecticides and transmission-blocking approaches. As research continues, the challenge will be to translate genomic insights into practical solutions while carefully considering their ecological and ethical implications. Ultimately, the study of the mosquito genome brings us closer to a future where malaria is no longer a global health threat.
Recent Posts
-
Genome of Anopheles gambiae: What We Know Today
Genome of Anopheles gambiae: What We Know Today Mosquitoes are among the most dangerous animals on E …24th Mar 2026 -
Analysis of Mosquito Immune Responses
Understanding Mosquito Defense Systems Against Pathogens Introduction Mosquitoes host complex microb …24th Mar 2026 -
CRISPR in Mosquito Control: Future or Risk?
CRISPR in Mosquito Control: Future or Risk? Mosquitoes are more than just annoying summertime pests …24th Mar 2026